|
O2-Which of the following would require the largest amount of energy to. So in the end we can conclude that nickel ions have a minimum radius. This is because for every number of protons it has it has an effect on its electrons. That is all three ions contain 18 electrons but. If you mean by atomic radius then helium has the smallest atomic radius. So the correct answer is Option C.Ĭorrect answer - Which of the following ions has the smallest ionic radius. L-ewwel teorija preċiża li tispjega n-natura. In periods, atomic radius decreases from left to right. Te CORRECT In groups, atomic radius increases from top to bottom. Which of the following has the smallest radius. Question 16 of 25 0.0/ 4.0 Points Of the following, which atom has the smallest atomic radius A. Which ion would you expect to have the smallest ionic. The Fish Tale Across the Wall Tenths and. B atom types C atom type We can say that the atom types make the atom types A and B the same. This means the atom types give different numbers of atom types. K Cl and S2 form an isoelectronic series with the Ar closed-shell electron configuration.īr- Ca2 K Se2- Physics. To do this, we have to assume that they are given the same number of atoms as the atom types. Which of the following ions has the smallest radius quizlet. Inside Our Earth Perimeter and Area Winds Storms and. Which of the following has smallest radius. Which of the following ions has the smallest radius. Be2 which of the following ions has the largest radius. Whic of the following ions has the smallest radius. Which of the following atoms and ions has the smallest radius. Which of the following pairs is isoelectronic. L-atomu huwa l-iżgħar partiċella ta element li għandu l-istess proprjetajiet kimiċi bħall-element tal-massa. Ca2 would have the smallest ionic radius because Calcium has a positive charge and. In general, the sodium atomic radius is 227 pm, while the aluminium atomic radius is 143 pm. So, an atom has no fixed radius, and the radii depend upon the type of bonds the atoms form. Ca2 would have the smallest ionic radius because Calcium has a positive charge and because this ion is a cation. Atomic radii are different depending on the type of bond an atom forms. On a banked race track the smallest circular path on which cars can move has a radius of 114 m while the.ġ O2- 2 S2- 3 Se2- 4 Te2. 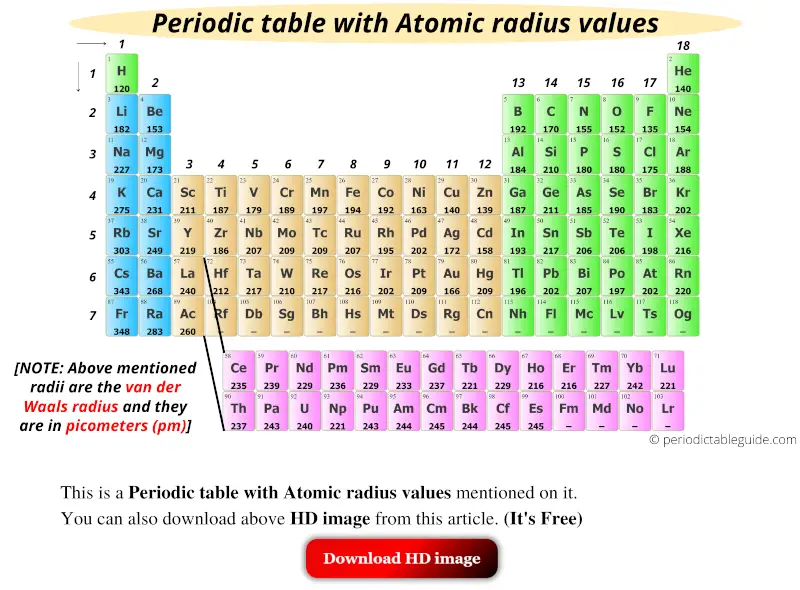 
While comparing the ionic radius we should consider all the points. 4 rows S2- K and Ca2 all have 18 electrons. Which ion would you expect to have the smallest ionic radius. Maps Practical Geometry Separation of SubstancesPlaying With Numbers India. We know that all the P block elements which is given in the options are belong to the same period and in the video as we go from left to right.Ĭlick here to get an answer to your question Which ion has the smallest radius. Arrange the following ions in order of increasing size. Periodic Trends Determine Which Atom Has The Smallest Atomic Radii Radius Johnny Cantrell Youtube A P B Cl- C Al D S2- E Ga So the answer is apparently A P. Which of the four iron has the smallest radius. I got a bit confused on why that is because I.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
